Rob Brown
Recent Posts
Batteries for Night Vision Goggles
[fa icon="calendar'] 12-Apr-2024 10:51:38 / by Rob Brown posted in Products & Services
It’s not a phone Nikita Kuzmin had in the Celebrity Big Brother house
[fa icon="calendar'] 21-Mar-2024 15:03:04 / by Rob Brown posted in Medical industry insight
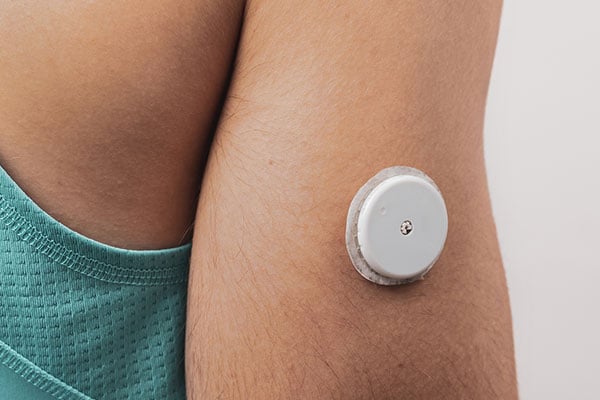
Ultralife manufacture Thin Cells® for CGM sensors and rechargeable batteries for handheld receivers, plus Thin Cells® can be used in insulin pump patches
The battery industry's most powerful women
[fa icon="calendar'] 08-Mar-2024 12:13:02 / by Rob Brown posted in Insider
March 8th is International Women's Day, so what better day to celebrate the contribution that women have made to the battery and charger industries?
Batteries can help the remote patient monitoring market to grow
[fa icon="calendar'] 15-Jun-2022 14:02:00 / by Rob Brown posted in Medical industry insight
~ Remote patient monitoring could grow by 128 percent but is the power sufficient? ~
Batteries for Smart Meters/Utility Meters
[fa icon="calendar'] 17-Mar-2022 14:17:00 / by Rob Brown posted in Products & Services
The first smart meter was developed and produced in the late 70s and, by the mid-80s, manufacturers of these devices found Lithium Thionyl Chloride (Li/SOCl2) cells to be an extremely well-suited source of power due to their long service life that reduces the need for replacement. Ultralife has recently introduced a new line of Li/SOCl2 cells, called ER Generation X, that are a great option for those who consider performance, price and lead time.
Managing festive fires in hotels
[fa icon="calendar'] 03-Dec-2021 12:04:25 / by Rob Brown posted in Safety and security industry insight
In hotels over Christmas, there is an increase in the use of electrical items (such as light up decorations), which may pose a risk of fires developing. Therefore, smoke alarm battery manufacturer Ultralife Corporation has put together an infographic offering a few fire safety tips to hoteliers, while emphasising the importance of installing proper smoke alarm batteries.
S is for Singh… and his views on project methodology
[fa icon="calendar'] 12-Nov-2021 10:13:00 / by Rob Brown posted in Project Management
When OEMs are undertaking new product development, asking the right questions and having a competitive advantage can mean the difference between success or failure. For instance, can improvements be made to ensure the products operate more effectively in their intended environments (particularly in industries such as medical and military)? Plus, with over 20 new laws and legislations being passed in the UK every year, there is the challenge to stay compliant in the process.
Is Finch movie preparing us to welcome robots into our family?
[fa icon="calendar'] 05-Nov-2021 11:26:45 / by Rob Brown posted in Robotics
Tom Hanks' 2021 film, Finch, tells of a man who is terminally ill and must teach his robot how to look after his dog when he's gone. The film conveys a possible family unit of tomorrow (a man, his dog and his robot – named Jeff – living in harmony).
Is your smart home device under the Christmas tree?
[fa icon="calendar'] 03-Nov-2021 15:18:00 / by Rob Brown posted in Products & Services
According to Amazon, a record number of smart home devices were sold during the 2018 holiday season, a trend that looks set to continue this year. However, sales are dominated by household names, such as Amazon and Google, making it difficult for competitors to secure a coveted place under the Christmas tree. Here, we look at a home automation and smart home security device from other manufacturers and discuss the ways that batteries are developing to meet the emerging power needs.
Search and rescue product development calls on batteries
[fa icon="calendar'] 14-Oct-2021 15:07:00 / by Rob Brown posted in Manufacturing industry insight
In an emergency, search and rescue devices need to be put swiftly to use and cannot shutdown until the mission is complete. Therefore, the source of power should be a vital consideration for OEMs early in the product development or design process. Taking emergency beacons and drones as examples; here we consider how the functionality of these devices can dictate the battery design.